 The electron donating tendency of metals reduces as we go down in the series. > Metals which are placed at the higher position can displace metals from their salt that are placed below to it. > The elements liberate hydrogen gas on reacting with dilute $HCl$ or dilute $$which are found above the hydrogen element in the reactivity series. > When we go down in the reactivity series the reducing ability and electro-positivity of elements decreases. > The metals which are placed at the top of the reactivity series are very good reducing agents as they are easily oxidized. The data provided by the reactivity series can be used to predict whether a metal can displace another in a single displacement reaction. For example-$Mg$ metal can displace hydrogen ions from its solution hence it is more reactive than element hydrogen.įeatures of the activity series of metals – The reactivity series of metals, also known as the activity series, refers to the arrangement of metals in the descending order of their reactivities. It also provides data on the reactivity of metals towards water and acids. In general, the more reactive a metal is: the more vigorously. Activity Series of Metals: The electrochemical, electromotive, or activity series of the elements is formed when the electrodes (metals and nonmetals) in contact with their ions are ordered on the basis of the values of their standard reduction potentials or standard oxidation potentials. The reactivity series provides information whether a metal can displace another in a single displacement reaction. The reactivity series of metals is a chart listing metals in order of decreasing reactivity. Now repeat steps 1-7 for the next three metals (Cu, Zn and Ag).Hint : We know that the activity series of metals is a list of metals where the arrangement of metals in the descending order of their reactivities. 
(Hint: these are single replacement reactions)Ĩ. 
In table 2, write the balanced equations for the reactions that occured. In data table 1, if a reaction occurred, write "reaction." If no reaction occurs write "no reaction".ħ. Click on each beaker to see the "molecular scale"Ħ. /classroom-resources/metals-in-aqueous-solutions-simulationĥ. Click on the link below or copy and paste it into your browser. To practice using the activity series chart in your reference tableġ. To practice writing single replacement reactions Based on Equation 1, aluminum is more active than copper and therefore replaces the copper (this is called a single replacement reaction). 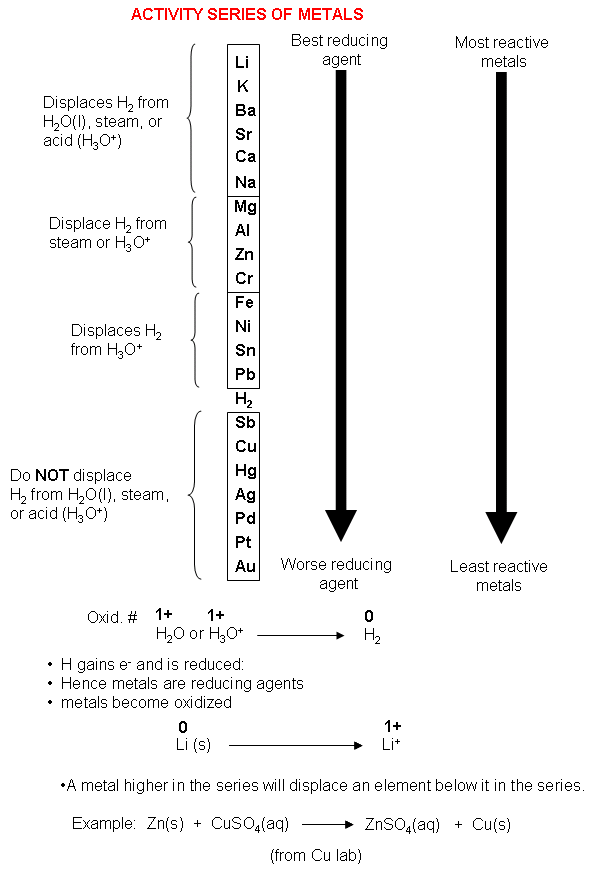
The more active metal forms a new compound containing metal cations. If it loses electrons really easily, it is really reactive, and so goes at the. In general, the activity of a metal may be defined as follows:Īn active metal will react with a compound of a less active metal, which is converted to its “free element” form. For metals, the reactivity of a metal depends on how easily it loses electrons. A more active metal always replaces the ion of a less active metal. Choose Activity 1, then choose Activity 4 to navigate to the metals + hydrochloric acid section. The activity or electromotive series of metals is a listing of the metals in decreasing order of their reactivity with hydrogen-ion sources such as water. If Al is above Co in the activity series of metals, which of the following will occur if Al metal is put into a solution of cobalt nitrate answer choices. Single replacement reactions will occur spontaneously in one direction only (compare Equations 1 and 2). swf ' file available on the right menu has the full activity series of metals computer simulation involving the following metals with HCl (aq): Ag, Cu, Fe, Mg, Ni, Pb, Sn, Zn. bubbles of oxygen gas will form on iron nail. This is the basis for single-replacement reactions. The reaction of aluminum with copper (II) chloride (Equation 1) is classified as a single replacement reaction – aluminum reacts with and “replaces” copper ions in copper (II) chloride. Now, looking at the activity series, we see that zinc is indeed more easily oxidized than hydrogen. Along the side of the matrix, you should list the five metals used in this part of the experiment: copper, iron, magnesium, tin, and zinc. Consider equation 1 and 2 below:ĢAl (s) + 3CuCl 2(aq) -> 2AlCl 3(aq) + 3Cu (s) (Equation 1)Ĭu (s) + AlCl 3(aq) -> No Reaction (Equation 2) To determine the activity of metals you can compare the reactions of metals with different How can we determine the relative reactivity of different metals? 
Gold is a highly valuable jewelry metal because it is essentially unreactive. Copper is used in electrical wiring because it conducts electricity extremely well and resists corrosion better than many metals. Although iron is the most common metal used in manufacturing, it must be protected againstĬorrosion because rusts easily. The usefulness of metals in structural and other applications depends on their physical and chemical 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
